A unique pharmaceutical development boutique
From concept to pharmaceutical Formulation and beyond
Avivia is a Dutch independent Pharmaceutical Contract Research Organization (CRO) located in the Netherlands (EU), and your partner in drug formulation, reformulation, repurposing, repositioning and
generation or protection of intellectual property.
Avivia is specialized in medicinal product development. From product evaluation to comprehensive formulation and analytical development, excipient characterisation, reverse engineering, dissolution test development,
stability studies, IMPD production, CMO selection and transfer.
Our expertise and capabilities are centred around formulation and CMC development of both generic drugs, life cycle management of branded drug products and new drug products (NCE).
Avivia has four complementary pharmaceutical R&D speciality service platforms:
Pharmaceutical R&D Services
Analytical R&D Services
Dissolution Lab Services
Excipia ™
Pharmaceutical R&D Services
Avivia offers pharmaceutical R&D formulation services, problem solving and troubleshooting, with small scale GMP batch manufacturing for a wide variety of medicinal dosage forms. Empower your drug discovery journey with our cutting-edge Pharmaceutical R&D Services. We offer a wide range of services to accelerate your product’s time to market.
Analytical R&D Services
Analytical Research and Development Services with pharmaceutical analysis experts who know what it takes to bring a pharmaceutical drug product to the market, from discovery, feasibility and transfer to GMP. Ensure the quality and safety of your pharmaceutical products with our Analytical R&D Services.
Dissolution Lab Services
An experience and knowledge-based dissolution method development platform that focuses primarily on supporting formulation development and problem solving. For us dissolution testing is never a routine test, but an essential tool in pharmaceutical formulation development. Optimize your formulation development with our Dissolution Lab Services.
Excipia ™
Excipia is an excipient characterisation and research service center, focusing excipient quality, formulation development support and reverse engineering or deformulation of pharmaceutical products. Our thorough, in-house developed, characterization protects your intellectual property and guarantees the stability and efficacy of your medicinal products.
Welcome to Avivia and we hope to be in touch soon!
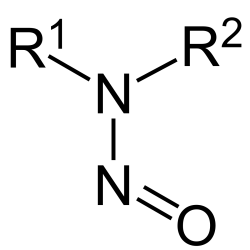
Nitrosamine prevention or reduction
Avivia advises and supports pharmaceutical companies and excipients manufacturers to mitigate and (re-)design drug product processes to prevent, reduce or eliminate nitrosamines. Avivia strives to provide solutions without affecting bioavailability and safety of the drug product and/or to minimize regulatory dossier variations.
Avivia for all
Big Pharma, speciality pharma, small entrepreneurial drug development firms, generic drug companies, universities, excipient manufacturers, law firms and even venture capital firms… our customer base is very broad and diverse. Although most of our activities are focused on wet-lab product development, we also provide hands-on support services in fields such as IP protection and development, investor’s due diligence, upscaling support, deformulation analysis, life cycle management strategy development, etc.
We have learned that serving many players with very different interests makes us stronger!
Be it the protection or creation of new formulation patents for originator products or the evasion of established formulation patents for generic drug development, we are constantly seeking new IP opportunities to maximize product value. Combined, our team are inventors on more than 30 granted patents on diverse drug products. Patents are in our DNA!
IP creation, protection and evasion
IP creation and evasion
Be it the creation of new formulation patents for originator products or the evasion of established formulation patents for generic drug development, we are constantly seeking new IP opportunities to maximize product value. Combined, our team are inventors on more than 30 granted patents on diverse drug products. Patents are in our DNA!
Avivia in numbers
Avivia to get you back on track
We like to get involved right from the start of a development project and work closely together with our clients and partners on transforming the idea into a product with a solid and commercially viable target product profile (TPP). We set the bar high but always work step by step with a clear focus on reaching the required end-result in the most efficient and cost-effective way possible.
Contact us and let’s see how we can work together on putting your ideas into practice!
Avivia to kick-start a development
In many of the projects we encounter, the development has derailed or – in the worst case and despite all previous efforts – has reached a dead end. We are known for our ability to see through the results and to bring up new ways of approaching the project leading to commercial success. Is it our product-driven mindset that makes the difference? Is it our interdisciplinary development approach or our unique technology capacities and experience?
Hard to say but we are very proud that we often succeed where others have failed before!
Network to accelerate
Avivia has access to a great team of pharmaceutical and analytical R&D professionals as well as to state-of-the-art laboratory infrastructure and equipment. We have also developed a large network or preferred partners in other fields such as pre-clinical development, GMP manufacturing and regulatory affairs. If relevant, we can always make use of their expertise and capacity to further optimize and accelerate customer projects.
It’s great to have friends!
Our core values
We are focused on
success
Success makes everybody proud and happy.
We are committed to
partnerships
Only by working closely together we can reach the mindset that leads to real and innovative products.
We are dedicated to the
highest standards
We always aim for the optimal result, fit to the situation!
“So, this is Avivia in a nutshell. I hope we have been able to trigger your interest so far! In the ‘Services’ and ‘Lab Capabilities’ sections of our website you can find the different services we offer to pharmaceutical, generics and drug development companies. If you are interested to learn more about Avivia, don’t hesitate to contact us and we would be happy to help you accelerating your development programs!”
JL, Managing Director Avivia BV



